Super-resolution dipole orientation mapping via polarization demodulation
- Select a language for the TTS:
- UK English Female
- UK English Male
- US English Female
- US English Male
- Australian Female
- Australian Male
- Language selected: (auto detect) - EN
Play all audios:

ABSTRACT Fluorescence polarization microscopy (FPM) aims to detect the dipole orientation of fluorophores and to resolve structural information for labeled organelles via wide-field or
confocal microscopy. Conventional FPM often suffers from the presence of a large number of molecules within the diffraction-limited volume, with averaged fluorescence polarization collected
from a group of dipoles with different orientations. Here, we apply sparse deconvolution and least-squares estimation to fluorescence polarization modulation data and demonstrate a
super-resolution dipole orientation mapping (SDOM) method that resolves the effective dipole orientation from a much smaller number of fluorescent molecules within a sub-diffraction focal
area. We further apply this method to resolve structural details in both fixed and live cells. For the first time, we show that different borders of a dendritic spine neck exhibit a
heterogeneous distribution of dipole orientation. Furthermore, we illustrate that the dipole is always perpendicular to the direction of actin filaments in mammalian kidney cells and
radially distributed in the hourglass structure of the septin protein under specific labelling. The accuracy of the dipole orientation can be further mapped using the orientation uniform
factor, which shows the superiority of SDOM compared with its wide-field counterpart as the number of molecules is decreased within the smaller focal area. Using the inherent feature of the
orientation dipole, the SDOM technique, with its fast imaging speed (at sub-second scale), can be applied to a broad range of fluorescently labeled biological systems to simultaneously
resolve the valuable dipole orientation information with super-resolution imaging. SIMILAR CONTENT BEING VIEWED BY OTHERS POLARIZATION MODULATION WITH OPTICAL LOCK-IN DETECTION REVEALS
UNIVERSAL FLUORESCENCE ANISOTROPY OF SUBCELLULAR STRUCTURES IN LIVE CELLS Article Open access 01 January 2022 POLCAM: INSTANT MOLECULAR ORIENTATION MICROSCOPY FOR THE LIFE SCIENCES Article
Open access 07 October 2024 QUANTITATIVE LINEAR DICHROISM IMAGING OF MOLECULAR PROCESSES IN LIVING CELLS MADE SIMPLE BY OPEN SOFTWARE TOOLS Article Open access 12 February 2021 INTRODUCTION
Through specific labeling of biological molecules, fluorescence microscopy plays an important role in visualizing subcellular organelles. The fundamental physical dimensions of fluorescence
include intensity (which reflects the fluorescence concentration), wavelength (absorption and emission spectrum), time (fluorescence decay lifetime) and polarization (which arises from the
dipole orientation). Because the fluorescent dipole orientation reflects the direction of the targeted protein, fluorescence polarization microscopy (FPM) has been studied extensively by
utilizing polarization modulation of excitation (linear dichroism)1, 2, 3 or analyzing fluorescence polarization (fluorescence anisotropy)4, 5, 6, 7 to measure the angle of a fluorophore—the
fluorescent dipole orientation—so that the direction of the targeted protein may be resolved. Unfortunately, these polarization-based investigations are limited by the optical diffraction
in conventional fluorescence microscopy. Super-resolution microscopy has been developed to extend our vision beyond the diffraction limit8. The key to achieve super-resolution is the
intensity On–Off modulation, either in a structured manner, such as stimulated emission depletion (STED)9, 10 or saturated structured illumination microscopy11, or in a stochastic manner,
such as (f) photo-activated localization microscopy (PALM)/stochastic optical reconstruction microscopy (STORM)12, 13, 14. These methods largely rely on intensity modulation using different
excitation wavelengths, often in association with temporal modulation. The wavelength dependency nature of these techniques critically requires the fluorescent labeling of a suitable
fluorophore, thereby limiting the applications of super-resolution microscopy15. In addition to fluorescence intensity, wavelength, and lifetime, the fourth dimension of
fluorescence—polarization—can also provide intensity modulation without the restriction to specific fluorophores; its investigation in super-resolution microscopy is still in its infancy.
Via fluorescent polarization modulation, Hafi _et al_16 recently developed a novel super-resolution technique through sparse deconvolution of polarization-modulated fluorescent images
(SPoD). Because the fluorescent dipole is an inherent feature of fluorescence, and its polarization intensity can be easily modulated with rotating linear polarized excitation, the
polarization-based super-resolution technique therefore holds great promise with regard to a wide range of biological applications due to its compatibility with conventional fluorescent
specimen labeling. Although super resolution can be achieved, the dipole orientation information is lost during SPoD reconstruction. Recently, there has been an interesting debate regarding
whether fluorescent polarization can be used to yield additional super-resolution information17, 18. Here, by fully exploring the advantages of fluorescent polarization, we present a new
technique called super-resolution dipole orientation mapping (SDOM) to extract the dipole orientation information beyond super-resolution imaging. In SDOM, a polarization-variant model is
established, in which the intensity determines the super-resolution microscopic image, while the phase determines the mutual dipole orientation of each super-resolved focal volume. The SDOM
algorithm uses sparsity-enhanced deconvolution to estimate effective dipole intensity under polarization modulation and applies least-squares estimation to extract the dipole orientation,
thus fully utilizing the polarization modulation information. The dipole orientation mapping, as a new dimension, can be superimposed onto the super-resolution image, in which the angle
denotes the effective dipole orientation, and the length denotes the orientation uniform factor (OUF), which describes the confidence of the orientation distribution of local dipoles. With
such valuable dipole orientation information, we reprocess the neural polarization modulation data set, and the membranes of the spine neck in the opposite direction show very different
polarization angles. This result clearly shows that the polarization does provide further structural information on top of the super-resolution image, thereby providing a timely answer to
the key question raised by the debate mentioned above. Moreover, by taking advantage of the parallel dipole orientation modulation with a low-excitation light level, we demonstrate that SDOM
performs at a temporal resolution as fast as 5 f.p.s., which allows live cell imaging to reveal the dynamic structural information of proteins. Because fluorescence polarization is widely
applied in different biological research areas, such as septin dynamics in live yeast cells6, 19, nuclear pore complexes20, 21, and protein structure and function in cell membranes1, 2, 22,
23, 24, 25, 26, our SDOM method bridges the gap between wide-field polarization imaging and super-resolution microscopy. We therefore anticipate widespread interest in and adoption of SDOM
as a powerful tool to answer a wide variety of key biological questions. MATERIALS AND METHODS EXPERIMENTAL DESIGN All organic fluorescent dyes and fluorescent proteins are dipoles, whose
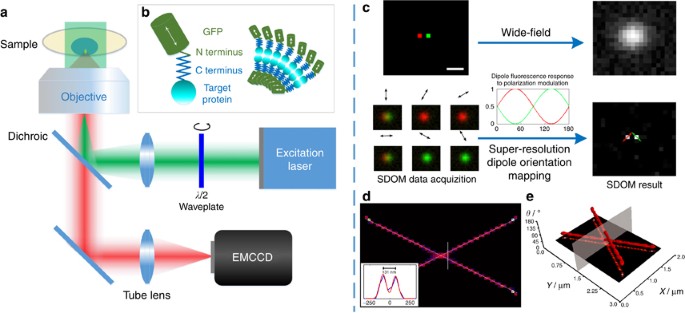
orientations are closely related to the structure of their labeled target proteins (Figure 1b)20. Because both the excitation absorption and fluorescence emission of dipoles have
polarization features, FPM has been widely used to study dipole orientation. SDOM is one type of FPM, working in the super-resolution region, and requires a series of fluorescence images
from a wide-field epi-fluorescence illumination microscope under different angles of polarized excitation. Polarized excitation can be achieved by using a rotary half-wave plate that is
synchronized to a camera (Figure 1a). There may exist multiple dipoles in the corresponding focal volume. Thus, the detected fluorescent anisotropic response is the integration of all of the
fluorescent molecules’ dipole orientation contributions. Unlike PALM/STORM12, 13, 3B27, SOFI28 and so on, in which intensity fluctuation is achieved stochastically, intensity fluctuation in
SPoD and SDOM is achieved through different dipole responses to polarized excitation; therefore, the demodulation algorithm is applied. In SDOM, effective dipole intensities under different
polarized excitations are estimated via sparsity-enhanced de-convolution16 of polarization data within the point spread function (PSF). The polarization modulation, described by a
cosine-squared (cos2) function, is then related to dipole orientation (Figure 1c) via least-squares estimation. Furthermore, we use OUF to evaluate the super-resolved dipole orientations.
Compared with the conventional FPM, where the neighboring fluorescent emitters cannot be discerned within the optical diffraction limit, SDOM, with super-resolution resolving power, analyzes
polarization data from several fluorescent emitters under polarization excitation modulation by analyzing the distinct periodic oscillating fluorescence signals (Figure 1c). The dipole
orientation (polarization information) map can be either superimposed on top of the super-resolution image or demonstrated in a (_X_, _Y_, _θ_) coordinate system. For example, if we simulate
two crossed lines with a dipole orientation difference of 90°, the image of orientation mapping and the (_X_, _Y_, _θ_) coordinate image will be as shown in Figure 1d and 1e, respectively.
By providing the polarization as an additional dimension to the fluorescence intensity, the nearby fluorophores can be clearly discerned by using SDOM, as shown in Figure 1c and
Supplementary Fig. S1. Figure 1d and 1e are the simulation results of two densely labeled intersecting lines with a uniform dipole orientation on each line. Figure 1d shows the orientation
mapping image of SDOM, with a resolution of ~130 nm, from the intensity information. Shown in a (_X_, _Y_, _θ_) coordinate system, two lines with different dipole orientations can be
completely separated. RECONSTRUCTION ALGORITHM OF SDOM The detected image _I_(_r_,_θ_) under illumination with a polarization angle _θ_ is subject to Poisson distribution, with the photons
reaching the sensor _μ_(_r_,_θ_) at each position _r_ and polarization angle _θ_: The _i_th emitting dipole at position _r__i_ is described by intensity _g_0(_r__i_) and orientation _α__i_.
The effective photons emitted from each dipole are modulated by the polarization angle of illumination with a cosine-squared function _f_(_θ,α__i_)=cos2(_α__i_−_θ_), which is blurred by the
PSF _U_(_r_) of the system. The entire imaging model can be described by Equation (2): Here, the background _b_(_r_) is modeled as a polarization invariant within a short time. _I_0(_θ_) is
a polarization-dependent periodic correction factor of the non-uniform response of the entire optical system. The purpose of the SDOM algorithm is to estimate both the intensity _g_0(_r__i_)
and the dipole orientation _α__i_. We denote _g_(_r__i_,_θ_) as the effective intensity under the illumination of polarization angle _θ_, which leads to
_g_(_r__i_,_θ_)=_g_0(_r__i_)_f_(_θ,α__i_). Thus, we can obtain Because the effective intensity _g_(_r__i_,_θ_) considers polarization modulation information, as suggested by Hafi _et al_16,
additional information is embedded in this model, which allows polarization super-resolution, benefitted also from a much sparser representation of the fluorescence intensity images. Maximum
a posteriori is applied to estimate _g_(_r__i_,_θ_) in this Poisson statistical model. _g_(_r__i_,_θ_) and b̃(_r_) are independently and identically distributed, respectively, with b̃(_r_)
being the cosine transform of _b_(_r_) to accelerate the computation16. The blurred background usually varies slowly in space, the property of which can be achieved by requiring the sparsity
of the cosine transform of _b_(_r_)16. Laplace (exponential) distribution can be applied to describe the prior sparsity of _g_(_r__i_,_θ_) andb̃(_r_)29. We maximize the maximum a posteriori
with and obtain the optimization model: argmin _L_(_g, b, I_), where After discretization, _L_ is a multivariate function consisting of a convex smooth part and a convex non-smooth part.
The fast iterative shrinkage-thresholding algorithm (FISTA) can be applied to achieve fast minimization30. By integrating _g_(_r__i_,_θ_) with respect to _θ_, the intensity _g_0(_r__i_) can
be obtained, which is equivalent to the SPoD super-resolution image. The orientation _α__i_ could be extracted from the polarization-variant effective intensity _g_(_r__i_,_θ_) using
least-squares curve fitting. Before that, because it is meaningless to calculate orientation in pixels without any dipoles, pixels containing no fluorescence signal can be marked on the
super-resolution intensity image _g_0(_r__i_). For pixels containing dipoles, each pixel could be affected by more than one dipole. Assume there are _n_ dipoles influencing pixel _i_, with
orientation α_j_(α_j_∈[0,π]) and the maximum number of photons _M__j_ reaching pixel _i_. The polarization-variant intensity could be expressed as: Through further calculation, Equation (6)
could be re-written in the form of Equation (7): For each pixel _i_, least-squares curve fitting could be applied to Equation (7) to estimate ᾱ_i_. Instead of obtaining the orientation of
each dipole, we calculated the ᾱ_i_, which represents the average dipole orientation in pixel _i_. Supplementary Fig. S3 illustrates the summation of multiple dipoles. Dipoles with similar
orientations show a strong mutual dipole, whereas when they distribute homogeneously the mutual dipole degrades such that it has a strong DC component (Supplementary Fig. S3a and S3b).
Adjusted-_R_2 is used to describe the quality of curve fitting and involves calculating _R_2 after normalizing the fitted data to [0, 1]. To guarantee the correctness of orientation mapping,
only pixels with relatively large adjusted-_R_2 are orientation-mapped on the image. Moreover, in Equations (6) and (7), _A_ and B have the following analytic forms: where _A_ is referred
to as the orientation amplitude, which contains the dipole orientation signal; and _B_ is referred to as the super-resolution translation, which contains the super-resolution signal. We
define OUF as OUF=A/B to evaluate our result, and from Equation (8) we have 0≤OUF≤1. OUF describes the orientation uniformity of dipoles within a PSF area. Simulation in Supplementary
Information describes how the divergence in orientation of dipoles can influence the OUF. In our orientation-mapped images, OUF is represented by the length of the arrow whose direction
indicates the dipole orientation. OPTICAL SETUP AND IMAGE ACQUISITION A 488-nm linear polarized continuous-wave laser (OBIS, Coherent, Santa Clara, CA, USA) is coupled to a
polarization-maintaining optical fiber that is installed on a Nikon Ti-E motorized system (Nikon, Tokyo, Japan). A half-wave plate (450–650 nm, Union Optics, Wuhan, China) is mounted to a
motorized rotary mount (G065117000, Qioptiq, Munich, Germany) to rotate the polarization of the incident laser. The rotation is monitored by using an infrared sensor and synchronized to an
electron-multiplying charge-coupled device (EMCCD, Evolva Delta 512, Photometrics, Tucson, AZ, USA). A 60 × oil-immersion objective (numerical aperture=1.4, ApoPlan, Nikon) is used for
epi-illumination and imaging. An additional 4 × relay lens (Nikon VM4X, Nikon) is used together with the Nikon 1.5 inner magnification (Nikon) to make the pixel size equal to 44.4 nm. The
excitation laser power is 20 W cm−2 for actin samples and 100 W cm−2 for live yeast samples. The exposure time of each image is 20 ms for actin samples and 100 ms for live yeast samples. The
rotation speed of the half-wave plate is adjusted for 10 acquisitions during a polarization modulation cycle of 180°. The total number of measurements is 10 for actin samples and 20 for
live yeast samples. SAMPLE PREPARATION The _Saccharomyces cerevisiae_ yeast strain AGY169-pRS416-ScCdc12-conGFP:GEN(4D4) was a generous gift from Prof. Amy Gladfelter’s lab; it has four
amino acids removed from the 3′ end of the septin Cdc12 and four amino acids removed from the 5′ end of the green fluorescent protein (GFP) with no linker between them. The _S. cerevisiae_
yeast strain, with GFP-tagged nuclear pore protein Nic96, was obtained from a GFP-tagged budding yeast protein library31 purchased from Invitrogen (Thermo Fisher Scientific, Waltham, MA,
USA). To obtain the series of polarized fluorescence images for SDOM analysis, _S. cerevisiae_ cells were grown in 3 ml of yeast extract-peptone-dextrose in 10-ml conical tubes and shaken at
30 °C for 10 h. The cells were refreshed for 2 h, and OD600 was checked before the cells were collected via 1000 rpm of centrifuge. Then, the collected cells were re-suspended in yeast
extract-peptone-dextrose, transferred to a slide, covered with a coverslip, sealed with VALAP and imaged. DATA ANALYSIS The length of each arrow, labeled on each pixel, is proportional to
the OUF value of each pixel. However, because the OUF of wide-field images is usually small, the lengths of arrows on these images are pre-magnified 2 × for easy visibility in this article.
To determine whether a pixel contains dipoles, a threshold based on global Otus’s method32 is used. This threshold is related only to which pixel should be fitted with dipole orientation and
could be adjusted accordingly, which has no effect on the dipole orientation or OUF calculated. The orientation mapped to the images of fluorescent beads, actin in fixed mammalian cells and
septin in _S. cerevisiae_ cells was calibrated carefully using polarizers and represents the true orientation of fluorescent dipoles. However, for images of a neuronal spine, we assumed
that the orientation parallel to the horizontal direction is 0° and that the clockwise direction is positive in our simulation. Thus, the polarization angles labeled in these results are
relative angles that may have a constant difference with real orientation. The source code and manual of SDOM, together with parameters and details for SDOM reconstruction can be downloaded
from: https://github.com/KarlZhanghao/SDOM. RESULTS AND DISCUSSION COMPARISON BETWEEN SDOM AND SPOD Through re-processing the data set presented in the SPoD method16, the comparable
super-resolution image using our SDOM method was obtained as shown in Figure 2a (right); no information is lost due to over-deconvolution compared with the reported image using the SPoD
method (Figure 2a, left). A significant advantage of SDOM is the achievement of a super-resolution image of fluorophore orientations. Zoomed-in SPoD and SDOM images of a dendritic spine neck
in hippocampal neurons were compared (Figure 2b and 2c). Arrows in the SDOM image are used to map the dipole orientation, and their lengths are proportional to OUF. With the powerful
capability of fluorophore orientation mapping, a heterogeneous distribution of dipole orientation on different borders of a dendritic spine neck can be revealed (Figure 2c), which may
suggest the presence of a double membrane structure and a difference in the morphology of the fluorescent molecule orientation on the opposite side of the neck. When both the fluorescence
intensity and fluorophore OUF are mapped using pseudocolor in the same image, we also find that the outer borders of the membrane usually have a larger OUF, while the inner parts have a
larger intensity. As shown in Figure 2e, the outer border is rendered as cyan by the large OUF, while the inner remains red (small OUF is not shown). This is consistent with the fact that
the orientations in the outer border are more uniform than those on the inner side because the fluorophores on the outer border are less influenced by fluorophores elsewhere. To determine
the validity of the arrows labeled in the SDOM images, the modulation data of fluorescence intensity with polarization direction could be observed in each pixel (Figure 2d) via our
purpose-programmed software (for details, see Supplementary Information). The fluorophore orientation can only be mapped when the adjusted-_R_2 of the sinusoidal fitting of modulation data
is sufficiently large. SDOM OF SIMULATED DATA AND FLUORESCENT BEADS Simulation is first performed on fluorescent beads, which are imaged thereafter to validate the SDOM method and to examine
the accuracy of the dipole orientation mapping of fluorophores. First, simulation of two neighboring emitters, separated by a series of distances from 50 to 150 nm with various dipole
orientations, is performed to test the super-resolution power of SDOM (Supplementary Fig. 1). We also generate a pattern of a 200-nm diameter circle for simulation (Figure 3a). In the
wide-field image, the circle looks solid, while the SDOM reconstructed image reveals a hollow circle shape. The reconstructed dipole orientation map clearly recovers the radial arrangement,
with a much larger OUF than the wide-field counterpart. We further image fluorescent beads with a diameter of ~500 nm in Figure 3b (F8888, Molecular Probes, Eugene, OR, USA). The rotational
symmetric distribution of orientation mapping of fluorescent dipoles on the surface of 500 nm beads is also consistent with previous studies (Figure 3b)16. Next, we verify the reliability of
the orientation mapping of SDOM super-resolved images. Two neighboring emitters with a distance of 100 nm are simulated in large numbers and statistically analyzed (Supplementary Fig. 2d).
After 100 repetitions, we can see from the diagram that the angle detection error is within 5° when two neighboring emitters have a 90° orientation difference. The angle detection error
becomes larger when the difference of orientation becomes smaller. Overall, the angle detection error is within 10° when the neighboring emitters have a dipole angle difference of ≥30°. SDOM
IMAGING OF ACTIN IN FIXED CELLS The actin of a mouse kidney tissue slice labeled with Alexa Fluor 568 phalloidin (F-24630, Molecular Probes, Eugene, OR, USA) is investigated using the SDOM
method; the fluorescence shows strong polarization. In the raw data, actin filaments of the same direction reach peak simultaneously, while filaments of different directions reach peaks at
different polarization angles of the incident laser. Figure 4a shows the average intensity of 10 different fluorescence polarization-modulated images, being equivalent to that in the
traditional wide-field image. Several neighboring filaments, indicated by arrows, could not be resolved by conventional microscopy but are distinguished by the super-resolved intensity image
of SDOM (Figure 4b). In the intensity profile of Figure 4b, two filaments with a distance of 240 nm can be separated by SDOM, with 82 and 80 nm FWHM of each filament, calculated by double
Gaussian fitting. Orientation mapping images using wide-field images (Figure 4c and 4e) and SDOM images (Figure 4d and 4f) are also compared. Because each single filament can be resolved in
the SDOM images, the dipole orientation calculated is mostly influenced by fluorophores on the local filament. Thus, the fluorescent molecules with the same dipole orientation, or an
isolated single-molecule, usually have a larger OUF, indicated by the lengths of the arrows. Because the OUFs in Figure 4c and 4e are very small, we have to magnify the OUF two-fold (which
is proportional to the lengths of arrows labeled in the image), compared with the OUF in Figure 4d and 4f, so that it can be shown properly. We also find that in the SDOM images, the dipole
orientation of fluorophores is mostly perpendicular to the direction of actin filaments, while in the wide-field images the orientation is nearly canceled by the different directions of
actin filaments. This fact is consistent with the definition of OUF and strongly demonstrates the importance of super resolution in fluorescence polarization imaging. SDOM IMAGING OF SEPTIN
PROTEIN IN LIVE YEAST CELLS To test how well SDOM can be used in live cell imaging, the GFP-labeled septin protein (Cdc12) in _S. cerevisiae_ cells6, 19, 33 is imaged. Septin is regarded as
the fourth type of cytoskeleton34 and shows strong fluorescence polarization as well. The cross-sectional view image (Figure 5a) and top-view image (Figure 5b) are orientation mapped with
both wide-field images and SDOM reconstructed images, both of which are consistent with PolScope results19 but with higher resolution and higher OUF. The typical double-ring structure of the
septin can be seen in the SDOM image; it cannot be distinguished in the wide-field image. The orientation mapped on the hourglass structure is consistent with PolScope results.19 To further
illustrate how well SDOM can perform to detect the nucleus structure of live cells, the nuclear pore complex protein (Nic96) of _S. cerevisiae_ cells, labeled with GFP, is also imaged using
SDOM (Figure 5c); its fluorescent anisotropy was reported elsewhere20. LIMITATION OF OUR WORK Although SDOM yields super-resolution information with orientation mapping, it has some
limitations. For the SDOM results of _S. cerevisiae_ live cells and the neuronal spine, some pixels containing a fluorescence signal cannot be mapped with orientation because the
adjusted-_R_2 is relatively small. Ideally, every pixel can be fitted with sinusoidal equations, even with a chaotic orientation distribution of dipoles within a PSF area. However,
non-mapping can happen when the dipole orientation rapidly changes at a scale similar to that of image acquisition time or a high level of system noise (such as polarization distortion of
the dichroic mirror, fluctuation of laser power and so on) buries the polarization modulation information. Moreover, in the SDOM algorithm, because the super-resolved orientation is mapped
in two steps, that is, estimation of dipole intensity followed by estimation of dipole orientation, the error of the intensity estimation may be transferred to the orientation estimation.
One-step orientation mapping may reduce the intermediate fitting error but will cause difficulty in formulating some criteria, such as OUF, to select robust orientations. Designing and
establishing other algorithms to solve the orientation or orientation distribution at the super-resolution level will be part of our future work. We should also note that the resolution of
SDOM is related to the local distribution of dipole orientations. SDOM performs well with samples having similar dipole orientations rigidly attached to the subcellular structure, thus
reflecting the underlying structure of the organelle. With densely labeled, homogenously orientated samples, SDOM can hardly resolve the polarization angle information due to the average of
dipoles. CONCLUSIONS In 1995, the 2014 Nobel laureate Prof. Eric Betzig35 proposed that super resolution can be obtained by separating neighboring molecules in different dimensions. Although
various super-resolution techniques have been demonstrated based on fluorescence intensity with respect to wavelength (STED/PALM/STORM) and lifetime (pulsed and time-gated STED), the
parameter of polarization, as the fourth dimension of fluorescence, has not been well-studied with respect to super-resolution techniques. However, fluorescent dipole orientation is an
important indicator of the underlying subcellular organelle structure and has been extensively studied in diffraction-limited microscopy, using fluorescence polarization techniques including
linear dichroism1, 2, 3 and fluorescence anisotropy4, 5, 6. The difference between the two techniques is that linear dichroism obtains the fluorescence polarization through excitation
modulation with linearly polarized excitation light, while fluorescence anisotropy is obtained through the detection of polarized emission from fluorescent dipoles. Because fluorescence
anisotropy is sensitive to the depolarization process, such as rotational diffusion or energy transfer2, linear dichroism yields more accurate results with respect to the structure and
functionality of the fluorescent molecules. In the past, fluorescence polarization was employed in the conventional microscopy setups, such as wide-field19, 36, confocal2, 3, two-photon
excitation1, 23, 37, 38, total internal reflection fluorescence25 and fluorescence recovery after photobleaching22, 26, to study protein structure and functions. Due to a large focal volume
being limited by optical diffraction, all these techniques suffer from the same problem, that is, too many fluorophores within the focal volume may cancel the dipole orientation information.
The results presented here by using the SDOM method show the great advantage of mapping the dipole orientation information in a super-resolved manner once the super-resolution and
fluorescence polarization methods work together. SDOM, using the same hardware setup and with advances in algorithms developed from its pioneering version, SPoD, developed by Hafi _et al_16,
provides additional super-resolved information regarding the dipole orientation of fluorophores and reveals the structure and functionality of targeted subcellular organelles with greater
detail. Here, we demonstrate that in both fixed cells and live cells, SDOM can yield dipole orientation information at super resolution. The fast imaging speed (at the second scale) also
makes it possible to observe the dynamics of bioprocesses in live cells. Compared with traditional orientation mapping with wide-field images, SDOM not only provides higher resolution images
but also fits the polarization-modulated data with a higher OUF, which makes the detection of orientation more robust to noise and the results more accurate with respect to local dipoles.
Moreover, we show that super-resolution dipole orientation can bring us to a new level of understanding regarding biological systems, such as the perpendicular structure of actin, the
radical orientation of the dipole on the Cdc12 septin protein and the inner-outer bilayer structure of the neuron axon membrane. In this paper, we have demonstrated the SDOM super-resolution
technique using both organic dyes and fluorescent proteins for a variety of biological specimens, such as a neuron, kidney slice and live yeast cell. It provides a completely new
dimension—polarization—to super-resolution microscopy. Furthermore, with dipole orientation information at sub-diffractional resolution and a speed of 5 f.p.s., we believe that SDOM will
attract immediate interest with regard to the super-resolution study of highly dynamic cellular processes. SDOM shows its advantages in comparing with the temporal resolution of PALM/STORM
(at the level of several minutes) and the spatial resolution of SIM (>150 nm), which are not sufficient to fulfill expectations, and the power of STED (∼100 MW cm−2) may potentially
inflict photodamage on the specimen. More importantly, SDOM is compatible with a wide range of conventional fluorescent dyes to yield super-resolution. REFERENCES * Lazar J, Bondar A, Timr
S, Firestein SJ . Two-photon polarization microscopy reveals protein structure and function. _Nat Methods_ 2011; 8: 684–690. Article Google Scholar * Kress A, Wang X, Ranchon H, Savatier
J, Rigneault H _et al_. Mapping the local organization of cell membranes using excitation-polarization-resolved confocal fluorescence microscopy. _Biophys J_ 2013; 105: 127–136. Article
Google Scholar * Wang X, Kress A, Brasselet S, Ferrand P . High frame-rate fluorescence confocal angle-resolved linear dichroism microscopy. _Rev Sci Instrum_ 2013; 84: 053708. Article ADS
Google Scholar * Axelrod D . Carbocyanine dye orientation in red cell membrane studied by microscopic fluorescence polarization. _Biophys J_ 1979; 26: 557–573. Article Google Scholar *
Weber G . Rotational Brownian motion and polarization of the fluorescence of solutions. In: Bailey K, Edsall JT, Anson ML editors. _Advances in Protein Chemistry_ VOL. 8. New York: Academic
Press. 1953, p 415–459. Google Scholar * Vrabioiu AM, Mitchison TJ . Structural insights into yeast septin organization from polarized fluorescence microscopy. _Nature_ 2006; 443: 466–469.
Article ADS Google Scholar * Sun MH, He HH, Zeng N, Du E, Guo YH _et al_. Characterizing the microstructures of biological tissues using Mueller matrix and transformed polarization
parameters. _Biomed Opt Express_ 2014; 5: 4223–4234. Article Google Scholar * Hao X, Kuang CF, Gu ZT, Wang YF, Li S _et al_. From microscopy to nanoscopy via visible light. _Light Sci
Appl_ 2013; 2: e108 doi:10.1038/lsa.2013.64. Article ADS Google Scholar * Hell SW . Microscopy and its focal switch. _Nat Methods_ 2009; 6: 24–32. Article Google Scholar * Yang X, Xie
H, Alonas E, Chen X, Liu Y _et al_. Mirror-enhanced super-resolution microscopy. _Light Sci Appl_ 2016; 5: e16134 doi:10.1038/lsa.2016.134. Article Google Scholar * Gustafsson MGL .
Nonlinear structured-illumination microscopy: wide-field fluorescence imaging with theoretically unlimited resolution. _Proc Natl Acad Sci USA_ 2005; 102: 13081–13086. Article ADS Google
Scholar * Betzig E, Patterson GH, Sougrat R, Lindwasser OW, Olenych S _et al_. Imaging intracellular fluorescent proteins at nanometer resolution. _Science_ 2006; 313: 1642–1645. Article
ADS Google Scholar * Rust MJ, Bates M, Zhuang XW . Sub-diffraction-limit imaging by stochastic optical reconstruction microscopy (STORM). _Nat Methods_ 2006; 3: 793–796. Article Google
Scholar * Hess ST, Girirajan TPK, Mason MD . Ultra-high resolution imaging by fluorescence photoactivation localization microscopy. _Biophys J_ 2006; 91: 4258–4272. Article Google Scholar
* Ding YC, Xi P, Ren QS . Hacking the optical diffraction limit: review on recent developments of fluorescence nanoscopy. _Chin Sci Bull_ 2011; 56: 1857–1876. Article Google Scholar *
Hafi N, Grunwald M, van den Heuvel LS, Aspelmeier T, Chen JH _et al_. Fluorescence nanoscopy by polarization modulation and polarization angle narrowing. _Nat Methods_ 2014; 11: 579–584.
Article Google Scholar * Frahm L, Keller J . Polarization modulation adds little additional information to super-resolution fluorescence microscopy. _Nat Methods_ 2016; 13: 7–8. Article
Google Scholar * Hafi N, Grunwald M, van den Heuvel LS, Aspelmeier T, Steinem C _et al_. Reply to "Polarization modulation adds little additional information to super-resolution
fluorescence microscopy”. _Nat Methods_ 2016; 13: 8–9. Article Google Scholar * DeMay BS, Bai XB, Howard L, Occhipinti P, Meseroll RA _et al_. Septin filaments exhibit a dynamic, paired
organization that is conserved from yeast to mammals. _J Cell Biol_ 2011; 193: 1065–1081. Article Google Scholar * Kampmann M, Atkinson CE, Mattheyses AL, Simon SM . Mapping the
orientation of nuclear pore proteins in living cells with polarized fluorescence microscopy. _Nat Struct Mol Biol_ 2011; 18: 643–649. Article Google Scholar * Mattheyses AL, Kampmann M,
Atkinson CE, Simon SM . Fluorescence anisotropy reveals order and disorder of protein domains in the nuclear pore complex. _Biophys J_ 2010; 99: 1706–1717. Article Google Scholar *
Fooksman DR, Edidin M, Barisas BG . Measuring rotational diffusion of MHC class I on live cells by polarized FPR. _Biophys Chem_ 2007; 130: 10–16. Article Google Scholar * Gasecka A, Han
TJ, Favard C, Cho BR, Brasselet S . Quantitative imaging of molecular order in lipid membranes using two-photon fluorescence polarimetry. _Biophys J_ 2009; 97: 2854–2862. Article Google
Scholar * Owen DM, Rentero C, Magenau A, Abu-Siniyeh A, Gaus K . Quantitative imaging of membrane lipid order in cells and organisms. _Nat Protoc_ 2012; 7: 24–35. Article Google Scholar *
Sund SE, Swanson JA, Axelrod D . Cell membrane orientation visualized by polarized total internal reflection fluorescence. _Biophys J_ 1999; 77: 2266–2283. Article Google Scholar * Velez
M, Axelrod D . Polarized fluorescence photobleaching recovery for measuring rotational diffusion in solutions and membranes. _Biophys J_ 1988; 53: 575–591. Article Google Scholar * Cox S,
Rosten E, Monypenny J, Jovanovic-Talisman T, Burnette DT _et al_. Bayesian localization microscopy reveals nanoscale podosome dynamics. _Nat Methods_ 2012; 9: 195–200. Article Google
Scholar * Dertinger T, Colyer R, Iyer G, Weiss S, Enderlein J . Fast, background-free, 3D super-resolution optical fluctuation imaging (SOFI). _Proc Natl Acad Sci USA_ 2009; 106:
22287–22292. Article ADS Google Scholar * Chang SG, Yu B, Vetterli M . Adaptive wavelet thresholding for image denoising and compression. _IEEE Trans Image Proc_ 2000; 9: 1532–1546.
Article ADS MathSciNet MATH Google Scholar * Beck A, Teboulle M . A fast iterative shrinkage-thresholding algorithm for linear inverse problems. _SIAM J Imag Sci_ 2009; 2: 183–202.
Article MathSciNet MATH Google Scholar * Huh W-K, Falvo JV, Gerke LC, Carroll AS, Howson RW _et al_. Global analysis of protein localization in budding yeast. _Nature_ 2003; 425:
686–691. Article ADS Google Scholar * Sezgin M, Sankur BI . Survey over image thresholding techniques and quantitative performance evaluation. _J Electron Imaging_ 2004; 13: 146–168.
Article ADS Google Scholar * Ong K, Wloka C, Okada S, Svitkina T, Bi E . Architecture and dynamic remodelling of the septin cytoskeleton during the cell cycle. _Nat Commun_ 2014; 5: 5698.
Article ADS Google Scholar * Mostowy S, Cossart P . Septins: the fourth component of the cytoskeleton. _Nat Rev Mol Cell Biol_ 2012; 13: 183–194. Article Google Scholar * Betzig E .
Proposed method for molecular optical imaging. _Opt Lett_ 1995; 20: 237–239. Article ADS Google Scholar * Abrahamsson S, McQuilken M, Mehta SB, Verma A, Larsch J _et al_. MultiFocus
Polarization Microscope (MF-PolScope) for 3D polarization imaging of up to 25 focal planes simultaneously. _Opt Express_ 2015; 23: 7734–7754. Article ADS Google Scholar * Ferrand P,
Gasecka P, Kress A, Wang X, Bioud FZ _et al_. Ultimate use of two-photon fluorescence microscopy to map orientational behavior of fluorophores. _Biophys J_ 2014; 106: 2330–2339. Article
Google Scholar * Vishwasrao HD, Trifilieff P, Kandel ER . _In vivo_ imaging of the actin polymerization state with two-photon fluorescence anisotropy. _Biophys J_ 2012; 102: 1204–1214.
Article Google Scholar Download references ACKNOWLEDGEMENTS This work is supported by the National Key Basic Research Program (973 Program, 2012CB316503), the National Instrument
Development Special Program (2013YQ03065102) and the National Natural Science Foundation of China (31361163004, 31327901, 61475010 and 61428501). MQZ was partially supported by UTD funds.
AUTHOR CONTRIBUTIONS KZ, LC and PX conceived the project. PX, JG and DJ supervised the research and designed the experiments. LC and KZ programmed the algorithm and analyzed the data. KZ,
XY, MW, LC and ZJ were primarily responsible for the key experiments. KZ, LC, PX, DJ and JG wrote the manuscript. All authors contributed to data analysis, discussions and manuscript
preparation. AUTHOR INFORMATION Author notes * Karl Zhanghao and Long Chen: These authors contributed equally to this work. AUTHORS AND AFFILIATIONS * Department of Biomedical Engineering,
College of Engineering, Peking University, Beijing, 100871, China Karl Zhanghao, Xu-San Yang, Miao-Yan Wang & Peng Xi * Department of Automation, Tsinghua University, Beijing, 100084,
China Long Chen, Zhen-Li Jing & Jun-Tao Gao * Bioinformatics Division, TNLIST, MOE Key Laboratory of Bioinformatics and Center for Synthetic & Systems Biology, Tsinghua University,
Beijing, 100084, China Long Chen, Zhen-Li Jing, Michael Q Zhang & Jun-Tao Gao * Department of Radiology, Peking University Third Hospital, Beijing, 100191, China Hong-Bin Han *
Department of Biological Sciences, Center for Systems Biology, The University of Texas, Dallas 800 West Campbell Road, RL11, Richardson, 75080-3021, TX, USA Michael Q Zhang * Department of
Basic Medical Sciences, School of Medicine, Tsinghua University, Beijing, 100084, China Michael Q Zhang * Institute for Biomedical Materials and Devices (IBMD), Faculty of Science,
University of Technology, Sydney, 2007, NSW, Australia Dayong Jin & Peng Xi Authors * Karl Zhanghao View author publications You can also search for this author inPubMed Google Scholar *
Long Chen View author publications You can also search for this author inPubMed Google Scholar * Xu-San Yang View author publications You can also search for this author inPubMed Google
Scholar * Miao-Yan Wang View author publications You can also search for this author inPubMed Google Scholar * Zhen-Li Jing View author publications You can also search for this author
inPubMed Google Scholar * Hong-Bin Han View author publications You can also search for this author inPubMed Google Scholar * Michael Q Zhang View author publications You can also search for
this author inPubMed Google Scholar * Dayong Jin View author publications You can also search for this author inPubMed Google Scholar * Jun-Tao Gao View author publications You can also
search for this author inPubMed Google Scholar * Peng Xi View author publications You can also search for this author inPubMed Google Scholar CORRESPONDING AUTHOR Correspondence to Jun-Tao
Gao. ADDITIONAL INFORMATION Note: Supplementary Information for this article can be found on the _Light: Science_ & _Applications’_ website . SUPPLEMENTARY INFORMATION SUPPLEMENTARY
INFORMATION (PDF 715 KB) RIGHTS AND PERMISSIONS This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party
material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons
license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
Reprints and permissions ABOUT THIS ARTICLE CITE THIS ARTICLE Zhanghao, K., Chen, L., Yang, XS. _et al._ Super-resolution dipole orientation mapping via polarization demodulation. _Light Sci
Appl_ 5, e16166 (2016). https://doi.org/10.1038/lsa.2016.166 Download citation * Received: 26 December 2015 * Revised: 09 May 2016 * Accepted: 17 May 2016 * Published: 21 October 2016 *
Issue Date: October 2016 * DOI: https://doi.org/10.1038/lsa.2016.166 SHARE THIS ARTICLE Anyone you share the following link with will be able to read this content: Get shareable link Sorry,
a shareable link is not currently available for this article. Copy to clipboard Provided by the Springer Nature SharedIt content-sharing initiative KEYWORDS * dipole * fluorescence
polarization microscopy * orientation mapping * polarization modulation * super-resolution
