A computational platform for high-throughput analysis of rna sequences and modifications by mass spectrometry
- Select a language for the TTS:
- UK English Female
- UK English Male
- US English Female
- US English Male
- Australian Female
- Australian Male
- Language selected: (auto detect) - EN
Play all audios:

ABSTRACT The field of epitranscriptomics continues to reveal how post-transcriptional modification of RNA affects a wide variety of biological phenomena. A pivotal challenge in this area is
the identification of modified RNA residues within their sequence contexts. Mass spectrometry (MS) offers a comprehensive solution by using analogous approaches to shotgun proteomics.
However, software support for the analysis of RNA MS data is inadequate at present and does not allow high-throughput processing. Existing software solutions lack the raw performance and
statistical grounding to efficiently handle the numerous modifications found on RNA. We present a free and open-source database search engine for RNA MS data, called NucleicAcidSearchEngine
(NASE), that addresses these shortcomings. We demonstrate the capability of NASE to reliably identify a wide range of modified RNA sequences in four original datasets of varying complexity.
In human tRNA, we characterize over 20 different modification types simultaneously and find many cases of incomplete modification. SIMILAR CONTENT BEING VIEWED BY OTHERS RNA MODIFICATIONS
DETECTION BY COMPARATIVE NANOPORE DIRECT RNA SEQUENCING Article Open access 10 December 2021 EXPANSION OF THE RNASTRUCTUROMEDB TO INCLUDE SECONDARY STRUCTURAL DATA SPANNING THE HUMAN
PROTEIN-CODING TRANSCRIPTOME Article Open access 25 August 2022 DETECTION TECHNOLOGIES FOR RNA MODIFICATIONS Article Open access 21 October 2022 INTRODUCTION RNA is an extensively modified
biological macromolecule. Over 150 chemically distinct modifications have been reported. The presence of methylated adenine, cytosine, and guanine in RNA was uncovered in the 1960s1, and
pseudouridine has been referred to as the “fifth base” for decades2. However, widespread interest in these epitranscriptomic marks has been raised by recent reports that underscore their
importance in a wide variety of developmental signalling. In stem cells the intracellular effector proteins SMAD2 and SMAD3 promote binding of the N6-methyladenosine (m6A) writer complex to
a subset of mRNAs associated with early cell fate decisions3. Likewise, a number of modifications are associated with disease. It has been demonstrated that the loss of taurine modification
in the anticodon of mitochondrial tRNA-Leu is responsible for mitochondrial myopathy, encephalopathy, lactic acidosis, and stroke-like episodes (MELAS)4. m6A is implicated in obesity5 and
associated with defects in functional axon regeneration in mice6. Aberrant methylation of cytosine-5 (m5C) in tRNAs has been linked to neuro-developmental disorders7. Recent interest in
epitranscriptomics has also been spurred by technical advances in next-generation sequencing (NGS) technology, which has allowed modifications in mRNA to be profiled individually. All of the
approaches based on Solexa/Illumina sequencing use antibodies to immunoprecipitate modified RNA, and/or apply chemical or enzymatic treatments to alter it and read out modifications as
mutations or truncations in the preparation of cDNA8,9. The primary caveat of these methods is that only a single type of modification can be profiled in each experiment, and specific
chemical, enzymatic and/or antibody reagents do not exist for every modification. Further complications can be caused by lack of specificity of the existing antibodies, in particular m6A and
m6Am10. Steps have been made towards uncovering modifications directly using long-read sequencing platforms11,12, but many technical challenges stand between these approaches and routine
use, not least a significant error rate in base calling13. NGS-based methods have also generated conflicting results in the past14,15, underscoring the need for orthogonal approaches. Mass
spectrometry (MS) is currently the only technique that can directly and comprehensively characterize chemical modifications in RNA sequences. The majority of RNA MS has focused on reducing
the RNA to mono-nucleosides and applying workflows analogous to metabolite analysis16. While these techniques are effective in determining what modifications are present in a sample, all
information about the location and co-occurrence of modifications is lost. This information is critical in complex samples to attribute modifications to specific RNAs. Even in simpler cases,
modification location and co-occurrence may be important for a phenotypic effect; for example, in microRNA 2′-_O_-methylation of the 3′-most nucleic acid sterically inhibits 3′ exonuclease
digestion, which increases the half-life of the modified microRNA in the cell17. For this reason there is an interest in analyzing samples in as close to their native states as possible.
Analysis of intact RNA oligonucleotides by tandem mass spectrometry (MS/MS) is capable of determining modification sites with single-nucleotide resolution, by comparing mass spectra with a
sequence database18. However, oligonucleotides are challenging to separate via mass spectrometry-compatible liquid chromatography (LC). The current approach of choice is reversed-phase
ion-pair liquid chromatography19. In addition to the experimental challenges, difficulties emerge in interpreting the acquired data. Considerable efforts toward automating data analysis have
been made in recent years, starting with SOS20 in 2002, Ariadne21 in 2009, OMA/OPA22 in 2012, and RNAModMapper23 in 2017, all of which are programs for database-matching or decoding the
complicated patterns of oligonucleotide fragmentation. However, none of the existing software solutions offers key features necessary to analyze data from large-scale experiments. First, no
software can efficiently handle the analysis of RNA oligonucleotide data—especially of more complex samples or involving many different modifications—in batch-compatible fashion. Second,
statistical validation strategies, such as false-discovery rate (FDR) estimation, are not implemented. This leads to unreliable sequence assignments and subjective manual assessment of
spectra for validation. Third, existing solutions do not tie into any larger analytical framework, making integration with other (e.g. quantitative) data difficult. In contrast, shotgun
proteomics has been sequencing peptides reliably for many years, and the inference, identification and quantification of proteins from constituent peptides has been automated to such a
degree that the field has matured into answering biological questions at a more fundamental level24. To fill this fundamental gap, we present a fast, scalable database-matching tool called
NucleicAcidSearchEngine (NASE) for the identification of RNA oligonucleotide tandem mass spectra. Our software is implemented within the OpenMS framework, an open-source toolset for
processing mass spectrometric data25. NASE will be fully integrated into the primary distribution of OpenMS in the upcoming version 2.5, and will then be available for download as part of
OpenMS at https://www.openms.de. In the meantime OpenMS builds containing NASE are available at https://www.openms.de/nase. Beyond speed and sensitivity, NASE provides advanced features like
FDR estimation, precursor mass correction, and support for salt adducts. Powerful visualization capabilities are available through OpenMS’ data viewer. By supporting the common interface of
The OpenMS Proteomics Pipeline26, NASE can be easily used in automated data analysis workflows. This interoperability also enables the label-free quantification of RNA oligonucleotides
based on NASE search results. Using four original datasets we demonstrate the capability of NASE to reliably identify a variety of RNA types from different sources, and show how data
visualization and label-free quantification can augment the interpretation of identification results. RESULTS RNA OLIGONUCLEOTIDE MS DATASETS Using nanoflow ion-pair liquid chromatography
coupled to high-resolution tandem mass spectrometry (nLC-MS/MS), we generated four datasets from RNA samples of increasing complexity. First, oligonucleotides with the sequence of mature
Drosophila let-7 microRNA, 21 nt in length, were produced synthetically in unmodified and modified (2′-_O_-methylated at the 3′ uridine) forms (“synthetic miRNA” dataset). We characterized a
1:1 mixture of both forms of this RNA. Replicate measurements were acquired using different normalized collision energy (NCE) settings in the mass spectrometer. Second, we prepared two
samples of an in vitro-transcribed yeast lncRNA (NME1, 340 nt long), one of which was treated with an RNA methyltransferase (NCL1) catalyzing the 5-methylcytidine (m5C) modification (“NME1”
dataset). Third, we used size exclusion chromatography to produce two samples containing long ribosomal RNAs (18S and 28S) from a human cell line (“human rRNA” dataset). Fourth, we generated
three biological replicates of human total tRNA from a cellular extract — a complex mixture of highly modified RNAs (“human tRNA” dataset). The “NME1”, “human rRNA” and “human tRNA” samples
were all digested with an RNA endonuclease (RNase T1) to generate oligonucleotide sequences of a length amenable to mass spectrometry. A POWERFUL SEARCH ENGINE FOR RNA MS DATA We developed
a sequence database search engine for the identification of (modified) RNA sequences based on tandem mass spectra. The software, termed NucleicAcidSearchEngine (NASE), was implemented within
the OpenMS framework and combines existing functionality (e.g. for data input/output, filtering, and FDR estimation) with newly developed features. (See Methods section for details.) Given
a mass spectrometry data file and a FASTA file containing target and decoy (shuffled or reversed) RNA sequences as inputs, NASE generates oligonucleotide-spectrum matches with statistically
meaningful FDR scores. OpenMS’ interactive viewer, TOPPView27, was extended to support RNA identification results obtained using NASE, mirroring and augmenting existing functionality for
visualizing peptide identifications in proteomics experiments. In addition to the built-in FDR calculation, NASE provides other features that set it apart from alternative tools that are
currently available. Even with extensive preparation, nucleotide samples frequently contain salt adducts (in the form of cations attached to the phosphate backbone). NASE searches can take
this into account, by allowing users to specify chemical formulas of adducts to consider in the precursor mass comparisons. Furthermore, NASE supports the correction of precursor masses for
MS2 spectra that were sampled from isotopologue peaks other than the monoisotopic one. Especially for longer sequences, MS2 precursor ions are often picked from higher-intensity, heavier
isotopologues by the mass spectrometer’s data-dependent acquisition software. Without adjustment, the precursor masses would not closely match the theoretical (monoisotopic) masses of the
correct oligonucleotides, leading to no assignment or incorrect matches. We implemented a correction that considers offsets corresponding to multiples of a neutron mass when comparing
precursor and oligonucleotide masses. This feature greatly increases NASE’s ability to identify oligonucleotides with longer sequences. Curiously, we observed cases where the instrument
software erroneously estimated the precursor (“selected ion”) m/z value to be below the apparent monoisotopic peak. We found that this could be corrected by allowing a negative offset (−1)
in the precursor mass correction. Finally, through the OpenMS toolbox NASE enables seamless label-free quantification of the oligonucleotides that were identified in a sample. A
corresponding analysis pipeline can be easily created and run using a graphical workflow editor. Supplementary Fig. 1 shows an example pipeline from our analysis of the NME1 data, using the
editor that is conveniently included with OpenMS28. MS-BASED SEQUENCING OF AN INTACT SYNTHETIC MICRORNA In our analysis of data from the synthetic miRNA sample, we found a strong dependence
of sequence coverage on the Normalized Collision Energy (NCE) value. Identical samples were run with NCE ranging from 5 to 55. The best results were obtained for an NCE of 20 (Supplementary
Fig. 2). Subsequent LC-MS/MS analyses, including of the NME1 and tRNA samples, were thus carried out with this NCE setting. At the optimal NCE, both unmodified and modified RNA were
detected, and the location of the modification could be determined with high confidence. 874 spectra were identified that passed our hyperscore cutoff, matching sequences of length 5–21 nt,
including the full-length let-7. The shorter sequences correspond to artefacts of incomplete solid-phase RNA synthesis, which are easily detectable by LC-MS. In the full 21-nt sequence we
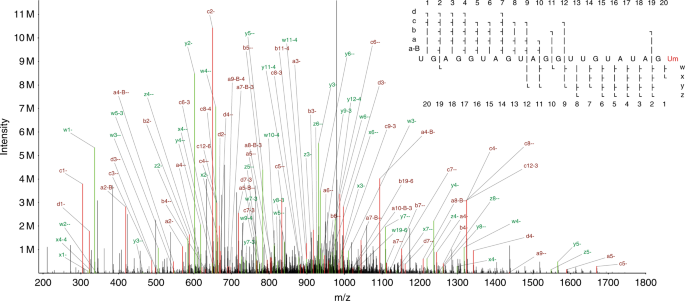
averaged over two-fold MS2 ion coverage of the let-7 sequence, with one or more forward (a-B/a/b/c/d) ion and one or more reverse (w/x/y/z) ion detected at each base (see Fig. 1, ion naming
scheme from McLuckey et al.29). This demonstrates our ability to sequence even relatively long (>20 nt) RNAs. PERFORMANCE COMPARISON OF SEARCH ENGINES FOR RNA MS DATA We processed the
NME1 data using the three search engines Ariadne, RNAModMapper, and NASE. We ran target/decoy database searches using m5C as a variable modification and combined results from all replicates.
We then compared the search engines in terms of: A, the number of identified spectra at different FDR thresholds; B, the sequence length distribution of the identified oligonucleotides at
5% FDR (Fig. 2). NASE identified significantly more spectra at a given confidence level than the other tools. It also found longer oligonucleotides, which would be more informative for
identifying RNAs in complex samples. About 10% of the oligonucleotide-spectrum matches generated by NASE at 1% FDR included sodium (2.9%) or potassium (7.2%) adducts and would have been
missed without the adduct search capabilities. Note that Ariadne’s performance in this comparison was hampered by the fact that a recommended tool for data preprocessing, the commercial
software SpiceCmd, was not available to us. RNAModMapper had previously been evaluated based on searches against “expected” sequences only (i.e. no decoys), followed by manual validation of
spectral assignments23,30. DETECTION OF DIFFERENTIAL METHYLATION SITES To assess the performance of our software at detecting RNA modifications, we compared the NASE search results for the
NME1 lncRNA with and without NCL1 incubation (Fig. 3a). Following common practice in the proteomics field31, we considered results at a high confidence level after filtering to 1% FDR and
removing “single hits” (oligonucleotides identified only based on a single spectrum) in each run. At this level, 72% and 73% sequence coverage were achieved for the control and the
NCL1-treated sample, respectively. As Fig. 3a shows, there is good agreement between the unmodified oligonucleotides that were identified in both samples, indicating that our method works
reproducibly. In the high-confidence set, m5C-modified oligonucleotides were only found in the NCL1-treated sample where they would be expected. Two illustrative examples are the isobaric
oligonucleotides “UCACAAAU[m5C]G” (at position 21-30 in the NME1 sequence) and “UAAC[m5C]CAAUG” (pos. 299-308) that were identified based on 29 and 10 spectra in multiple charge states (−2
to −5). Two additional identifications were made of the sequence “UAACC[m5C]AAUG”, i.e. with a shifted localization of the modification. Figure 4a shows a corresponding data section from the
NCL1-treated sample, visualized as a two-dimensional LC-MS map. Identifications of the unmodified, adducted, and modified variants of the two oligonucleotides are displayed in the context
of MS1 signal intensities. At the bottom, “UCACAAAUCGp” (left) and “UAACCCAAUGp” (right) can be seen eluting in overlapping peaks. (In our notation, “p” at the end of a sequence represents
the 3′ phosphate generated by RNase T1 cleavage.) In the middle, the corresponding mono-methylated oligonucleotides are convincingly detected, with a mass shift of 14 Da and a slight RT
shift relative to their unmethylated counterparts. At the top, the unmodified oligonucleotides were identified with a sodium adduct (mass shift of 22 Da). A corresponding image showing the
loss of signal for the modified oligonucleotides in the control sample is available as Supplementary Fig. 3. In Fig. 4b we compare spectrum matches for the two modified oligonucleotides,
showcasing the high quality of the matches as well as our MS2 visualization capabilities, including the newly added ion coverage diagrams. LABEL-FREE QUANTIFICATION OF RNA MS DATA We
quantified the identified oligonucleotides in the two NME1 samples, using a label-free, feature detection-based approach. Figure 3b summarizes the results. Although all oligonucleotides come
from the same RNA, they were quantified with signal intensities spanning several orders of magnitude. This is indicative of widely varying ionization efficiencies during MS analysis, a
common caveat that generally limits label-free quantification to relative comparisons between similar samples. Of 26 and 36 different oligonucleotide sequences that were identified as part
of the high-confidence set in the control and NCL1-treated sample, respectively, 25 and 32 could be quantified in either sample based on at least one replicate (corresponding to 96% and 89%
success rates). Unmodified oligonucleotides were quantified at similar levels in both NME1 samples, with a correlation of 0.94 of the log-intensities per sample (median of the technical
replicates) for oligonucleotides in all charge/adduct variants (0.98 when considering only the “best” variant in terms of reproducibility across replicates, as measured by the coefficient of
variation). Methylated oligonucleotides were only identified and quantified in the NCL1-treated sample, but their unmodified counterparts exhibited lower feature intensities in the treated
sample compared to the control, consistent with a partial shift of the ion current to the modified variants (Supplementary Fig. 4). More advanced capabilities for LC-MS-based quantification,
including retention time alignment, inference of identified analytes across samples, and labelling approaches, are already available in OpenMS for proteomics experiments. With future
improvements to the support for nucleic acids in the framework, these features will become available for RNA analyses as well. COMPARATIVE ANALYSIS OF HUMAN RIBOSOMAL RNA Taoka et al.
recently published a complete landscape of the modifications on human ribosomal RNA in TK6 cells32. We acquired LC-MS/MS data on human rRNA in HAP1 cells, analyzed it using NASE and compared
our results to their published findings (see Supplementary Data 1). For this comparison we filtered spectrum matches to 5% FDR and removed “single hits”; where oligonucleotides with the
same raw sequence were identified in different modification states (incl. unmodified), only the one with highest spectral count was considered. Our approach achieved 39% sequence coverage
for 18S rRNA and 25% for 28S rRNA, which is far from complete, but much higher than the coverage achieved by Taoka et al.32 using Ariadne on comparable data (RNase T1 digest; 13% for 18S and
7% for 28S). Using NASE we identified more and longer oligonucleotides; in the Ariadne data, almost a third of the oligonucleotides are short and match in several locations in the rRNA,
making them unsuitable for modification mapping without additional information. Taoka et al.32 reported 68 post-transcriptional modifications (not counting pseudouridines) on 18S and 28S
rRNA in regions where we have sequence coverage. For 57 of these modifications (84%) our identifications agreed exactly. In addition, five modifications were mis-localized by one position
and two by two positions in our results. In three of the remaining four cases, it is plausible that the unmodified ribonucleotides were correctly detected, because the identifications are
supported by high spectral counts (26, 39, 170) and the modification stoichiometry is not expected to be 100% according to Taoka et al.32. We further identified 10 modifications based on six
oligonucleotides that are not supported by the published data, with half of these coming from only two multi-modified oligonucleotides. The oligonucleotides were all found with low spectral
counts (2–4) and likely constitute false positives—with one exception, the sequence “AUC[mG]CCCCAG” which matches in 28S rRNA and was identified 25 times. It would be an interesting
candidate to investigate for cell line-specific differences in rRNA modifications. ANALYSIS OF A COMPLEX, HIGHLY MODIFIED TRANSFER RNA SAMPLE Previous work on tRNA has shown that it is
heavily modified33. Our analysis confirms this. We ran NASE on the “short RNA” fraction of a cell extract sample that had been digested with RNAse T1. We searched for 23 variable
modifications with different molecular masses, which had previously been identified to be present in yeast or human tRNA34,35. Most of these represent sets of isobaric modifications which we
cannot distinguish, such as position-specific variants of the same modification; e.g. “mC?” was used to represent any singly-methylated adenosine (incl. Cm, m5C etc.). Note that it was not
feasible to search this dataset with this high number of variable modifications using other available database-matching tools (RNAModMapper, Ariadne). After filtering to 5% FDR and keeping
only sequences that were found in the majority of replicates, the effective FDR in the dataset was 2.7%. At this level 13,654 spectra were matched to 304 different oligonucleotides. The
sequences of human tRNAs are highly similar, especially for tRNAs of one isotype, i.e. tRNAs that bind the same amino acid. Consequently, only 48 (16%) of the identified oligonucleotides map
to a unique tRNA sequence; however, 234 (77%) and 289 (95%) map uniquely to a single tRNA isoacceptor (same anticodon) or isotype, respectively. Considering only the oligonucleotides that
map uniquely to a specific isoacceptor, the highest sequence coverage was achieved for tRNA-ArgTCG (Fig. 5a). Coverage levels along the tRNA sequences were far from uniform, with the
majority of identified oligonucleotides overlapping the anticodon loop and 3′ anticodon stem, or the T-loop and 3′ T-stem (Fig. 5b). We hypothesize that the corresponding parts of the tRNA
structure are more amenable to RNase T1 digestion than other regions. Many of the oligonucleotides we identified contained multiple modifications. In the search, up to three modifications
per oligonucleotide were allowed, to limit the combinatorial space of modified sequences that needed to be explored. Of the unique oligonucleotides identified, 12% were unmodified
(accounting for 21% of the identified spectra), while 35% carried one, 28% carried two, and 25% carried three modifications (accounting for 32%, 31% and 16% of the identified spectra,
respectively). All modifications considered in the search were detected as part of identified oligonucleotides. However, the prevalences of different modifications differed widely—see Table
1 for details. Existing data on the modification landscape of human cytosolic tRNAs is incomplete (e.g. MODOMICS lists information for 36 tRNAs covering 16 isotypes) and at least some
modifications are differentially regulated, complicating comparisons. We will focus on cytosine monomethylation (mC, represented by “mC?” in our search) as one example that has been studied
more thoroughly, e.g. via bisulfite sequencing to detect m5C. We identified 36 unique oligonucleotides containing one (32) or two (4) mC site(s), based on a total of 1728 matched spectra. In
all, 30 oligonucleotides mapped uniquely to a single tRNA isoacceptor (codon); a further four mapped to a single tRNA isotype (amino acid) but multiple isoacceptors. The two remaining
oligonucleotides “AUU[mC]CAG” and “ADU[mC]CAG” could have come from either tRNA-ArgACG (pos. 46-52) or tRNA-Tyr (pos. 58-64); however, for tRNA-Tyr a conserved methylation (m1A) at A58 would
be expected, making tRNA-ArgACG the more plausible origin. Excluding this ambiguous case, at the level of isotypes a total of 19 unique mC sites were identified. Seven of these sites agree
with the “canonical” m5C sites in the VL junction of tRNAs at consensus sequence positions 48–507. Other common mC sites in tRNAs are pos. 32 and pos. 34, the wobble position in the
anticodon. We observed mC32 for tRNA-Arg (m3C reported in MODOMICS) and tRNA-Gln (Cm reported), and mC34 in tRNA-Met (Cm reported) and tRNA-Leu (m5C reported). In addition, we identified the
oligonucleotide “A[mC]U[mC]CA[mG]” a total of 35 times, which matches at pos. 31-37 in tRNA-Trp and implies mC at both pos. 32 and 34, as well as mG at pos. 37. No data for tRNA-Trp is
available in MODOMICS. Beyond methylcytidine, known recurring modifications that we identify in several tRNAs include monomethylation at G10, dihydrouridine at U20, mono- or dimethylation at
G26, N6-threonylcarbamoyladenosine (t6A) at A37 and monomethylation at A58. Interestingly, we observe 5-methoxycarbonylmethyl-2-thiouridine (mcm5s2U) at the wobble U34 of tRNA-ArgTCG/T,
tRNA-GlnTTG, tRNA-GluTTC and tRNA-LysTTT; this specific modification has been characterized in cow (tRNA-ArgTCT) and rat (tRNA-GluTTC and tRNA-LysTTT), but has not been directly located in
human samples36. This modification, which is installed by three consecutive enzymatic steps, has been reported to be induced in oncogenic conditions, and is important for tuning the
expression of protein factors based on their codon content37. The ability to identify and localize multiple different modifications simultaneously is an unique advantage of the
oligonucleotide MS approach. In many cases we find additional, alternatively modified (or unmodified) variants of “expected” oligonucleotides. In particular, for an oligonucleotide that
matches the T-loop region in several tRNA-Ala genes we robustly detect the doubly methylated form (mU55 and mA58), both singly methylated forms and the unmodified form. For the equivalent
oligonucleotides in tRNA-CysGCA, we found at least 60 matches for each of the double methylation and a single methylation at A58. In oligonucleotides overlapping the anticodon loop and the
3′ anticodon stem, we detect multiple forms e.g. for tRNA-GluCTC (unmodified and mC39), tRNA-GlyCCC (mU39 with and without mU32), tRNA-Met (either or both of mC34 and t6A37) and tRNA-iMet
(unmodified and t6A38). In tRNA-GluTTC we find mcm5s2U as well as its precursor mcm5U at U33 (presumably mis-localized from the wobble U34). For tRNA-Ser we observe several different forms
in this region—primarily mA37 and t6A42 with or without mU44 for tRNA-SerGCT, and N6-isopentenyladenosine (i6A) at A37 with either, both or neither of mU39 and mU44 for tRNA-SerA/CGA. In
tRNA-LysTTT, among a number of identified oligonucleotides all covering pos. 31–42, the four with highest spectral counts (all above 25) show what could be interpreted as a modification
cascade: first t6A at pos. 37, then addition of mcm5U at pos. 34, followed by conversion to mcm5s2U at pos. 34, and finally conversion to ms2t6A at pos. 37; see Fig. 6 for annotated spectra.
Based on our data alone it is impossible to determine whether these and other cases correspond to partial modifications of a particular tRNA, or to mixtures of differently modified tRNAs
from separate genes. However, overall these results support newer findings that question the stoichiometric and static nature of tRNA modifications, and favor the notion of a complex and
dynamic tRNA modification landscape38. DISCUSSION NASE is an open-source database search engine for RNA, optimized for high-resolution MS data. It supports arbitrary modifications, salt
adducts, and FDR estimation through a target/decoy search strategy. Moreover, integration with the OpenMS toolbox enables high-quality data visualization, e.g. for manual validation of
spectral assignments, and label-free quantification of RNA oligonucleotides. We have tested NASE against a range of sample types and complexities, spanning synthetic nucleic acids, in
vitro-transcribed sequences, and cell extracts. In all of these experiments we have been able to effectively identify RNA sequences and their modifications. NASE contains many unique
functionalities that are not currently realized in other database search tools for RNA. To our knowledge, no other tools account for precursor mass defect resulting from instrumental
selection of higher isotopologue peaks. This functionality is a major contributor to the excellent performance of NASE in identifying longer oligonucleotides compared to other
database-matching tools. NASE also provides powerful correction for cation adduction events, which lessens the impact of sodium and potassium ions on sequence characterization. In addition,
OpenMS in general and NASE specifically were designed to be fast. Our search times for complex samples are orders of magnitude faster than other tools. The searches on the NME1 and let-7
data take seconds; the much more complicated 23-modifications searches of the tRNA dataset took <30 min per file on our server (using 40 parallel threads). For comparison, an analogous
search using RNAModMapper was not feasible, with an estimated running time of one month. An equivalent search with Ariadne did not return any modified oligonucleotides. The open-source
nature of OpenMS and NASE enables users to modify the software to fit their specific needs, to extend the existing functionality, and to create new interoperating programs. Already, many
analysis tools have been implemented within the OpenMS framework to support mass spectrometry-based proteomics and metabolomics experiments. The present work, and here in particular the
pioneering application of label-free quantification, gives a foretaste of the power of leveraging these methods for the analysis of nucleic acid data. Future developments will streamline the
use of OpenMS tools and algorithms, e.g. for improved quantification and comparisons across many samples, in the field of epitranscriptomics. In conclusion, the development of NASE is an
important step towards the large-scale analysis of RNA by mass spectrometry. METHODS LIQUID CHROMATOGRAPHY-TANDEM MASS SPECTROMETRY RNA samples were separated by reversed-phase ion-pair
liquid chromatography (using 200 mM HFIP + 8.5 mM TEA in H2O as eluent A, and 100 mM HFIP + 4.25 mM TEA in methanol as eluent B) and characterized by negative ion MS/MS in a hybrid
quadrupole-orbitrap mass spectrometer (Q Exactive HF, Thermo Fisher). A gradient of 2.5 to 25% eluent B eluted oligonucleotides from various lengths of nanoflow Acclaim PepMap C18 solid
phase (Thermo Fisher) at 200 nL per minute. The length of the gradient was varied according to the complexity of the sample. Precursor ion spectra were collected at a scan range of 600–3500
m/z at 120k resolution in data-dependent mode, with the top five MS1 species selected for fragmentation and MS2 at 60k resolution. RNA SAMPLES A variety of RNA samples were characterized by
nanoflow LC-MS/MS (nLC-MS/MS) and sequence analysis performed using NASE. Initial work was carried out on a mature _Drosophila_ let-7 sequence that was prepared by solid-phase synthesis and
purchased from IDT. This sequence is a 21 nt long microRNA that was among the first miRNAs to be characterized39. The RNA was chemically synthesized in unmethylated and methylated forms,
i.e. with or without a 2′-_O_-methyluridine (Um) at position 21. A sample was prepared by mixing both forms, and was characterized by nLC-MS/MS without further processing, but with varying
normalized collision energy (NCE) settings to give different levels of precursor fragmentation. Subsequent experiments were carried out on NME1, a 340 nt long _Saccharomyces_ lncRNA. NME1
RNA was generated by in vitro transcription, and two samples with and without NCL1 enzyme treatment were prepared. NCL1 is a yeast RNA methyltransferase that catalyzes the 5-methylcytidine
(m5C) modification40. RNA was extracted and digested with RNase T1; this endonuclease produces shorter oligonucleotides by cleaving immediately after guanosine residues. nLC-MS/MS analysis
of technical triplicates of 100 ng of oligonucleotides was performed. For the human ribosomal RNA dataset, total RNA was extracted from HAP1 tissue culture by using Qiazol reagent according
to the manufacturer’s instructions. Samples were generated by size-exclusion chromatography of total RNA.41 Briefly, total RNA was fractionated through an Agilent Bio SEC-5 column using a
mobile phase of 100 mM ammonium acetate (pH 5) at a flow rate of 250 uL per minute. Two fractions containing long ribosomal (18S and 28S) RNA were digested with RNase T1 and 250 ng of
oligonucleotides were subsequently analyzed by nLC-MS/MS in technical triplicate. The most complex sample was a solution of digested crude human cellular tRNA, which was isolated in three
biological replicates from HAP1 tissue culture using an RNeasy kit (Qiagen) according to the manufacturer’s instructions. Briefly, RNAs can be fractionated by length by differential elution,
with RNAs less than 200 nucleotides mostly made up of tRNA, and the larger fraction being mostly rRNA. The “shorter” RNA fraction was digested with RNase T1, and the resultant
oligonucleotides were characterized by nLC-MS/MS in technical triplicate, with 100 ng being injected each time. NASE IMPLEMENTATION NASE was implemented in C++ within the OpenMS framework.
The OpenMS library was extended with classes representing (modified) ribonucleotides (based on data from the MODOMICS database42), RNA sequences, and endoribonucleases. A generalized data
structure for spectrum identification results (supporting peptides/proteins, nucleic acid sequences, and small molecules) and an algorithm for theoretical spectrum generation of RNAs were
added as well. NASE itself is an executable tool that supports the common interface of The OpenMS Proteomics Pipeline26. Data processing with NASE works as follows: Inputs are an RNA
sequence database (FASTA format) and a mass spectrometry data file (mzML format). RNA sequences are digested in silico using enzyme-specific cleavage rules for the user-specified RNase.
Tandem mass spectra are pre-processed (intensity filtering, deisotoping) and mapped to oligonucleotides based on precursor masses. Mass offsets due to salt adducts or precursor selection
from heavier isotopologue peaks can be taken into account. Next, theoretical spectra of relevant oligonucleotides in the appropriate charge states are generated and compared to the
experimental spectra; matches are scored using a variant of the hyperscore algorithm43. If the sequence database contains decoy entries, the resulting oligonucleotide-spectrum matches can be
statistically validated through the automatic calculation of _q_-values, a measure of the FDR44. Supported output formats are an mzTab-like45 text file, suitable for further analysis, and
an XML file, suitable for visualization in TOPPView. In order to provide support for label-free quantification of identified oligonucleotides, NASE interfaces with the OpenMS tool
FeatureFinderMetaboIdent (FFMetId). FFMetId handles the core step of the quantitative workflow, the detection of chromatographic features in the LC-MS data. As a variant of the proteomics
tool FeatureFinderIdentification46, FFMetId provides targeted feature detection for arbitrary chemical compounds. NASE can write an output file with all relevant information about the
oligonucleotides it identified, which is directly suitable as an input file for FFMetId. SEQUENCE DATABASE SEARCHES For NASE analyses, all proprietary raw files were converted to mzML
format47 without compression and with vendor peak-picking using MSConvert48 (https://github.com/ProteoWizard). The full list of fragment ion types (a-B, a, b, c, d, w, x, y, z) was
considered for peak matching. Precursor and fragment mass tolerance were both set to 3 ppm. For the synthetic let-7 data, an extensive set of potential adducts (Na+, K+, Na22+, K22+, NaK2+,
Na33+, K33+, Na2K3+, NaK23+) was used during the search because of the substantial salt that remained from the RNA synthesis reactions. Two copies of the let-7 sequence, one with a fixed
2′-_O_-methylation of uridine (Um) at the 5′ position, were specified in the FASTA sequence file. The small size of the sequence database prevented the use of a target/decoy approach for FDR
estimation. We thus used a stringent hyperscore cutoff of 150 (corresponding to the 1% FDR in the tRNA sample, see below) to define a high-confidence set of results. In the three other
datasets, results from target/decoy database searches were initially exported from NASE at 10% FDR (spectrum match-level), then further filtered in post-processing depending on the analysis.
For the NME1 data, the sequence database contained the NME1 (target) sequence as well as a shuffled decoy sequence. For the human rRNA data, to allow direct comparison with published
results, we used a sequence database containing 18S and 28S rRNA from TK6 cells (plus reversed decoys). A set of seven variable modifications, including monomethylations of all four
canonical ribonucleotides, was defined based on known ribosomal PTMs. In our search of the tRNA data, 23 variable modifications (based on reported modifications in human tRNA36) were
specified, at a maximum of three modifications per oligonucleotide. The FASTA file contained 420 human tRNA sequences collected from the tRNA sequence database tRNAdb35
(http://trna.bioinf.uni-leipzig.de) plus the same number of reversed decoy sequences. See Table 2 for additional dataset-specific parameters. SEARCH ENGINE COMPARISON The NME1 data was
processed with two other publicly available RNA identification engines, in addition to NASE: Ariadne21 and RNAModMapper23. To this end, the raw files were converted to MGF format using
MSConvert. Cleavage and variable modification settings in the searches were the same as for NASE and appropriate for the samples. For Ariadne, the online version at http://ariadne.riken.jp
was used in July 2019. The “Calc as partial modifications” option was enabled. The precursor and fragment mass tolerances were left at their default values (5 and 20 ppm). Alternatively,
using the parameters from the Taoka et al.49 (20 and 50 ppm) made no appreciable difference for Ariadne’s performance in our tests. For RNAModMapper, a program version from July 2018 was
used with settings recommended by the author, Ningxi Yu. All available ion types (a-B, w, c, y) were enabled; precursor and fragment mass tolerances were set to 0.02 and 0.1 Da,
respectively. LABEL-FREE QUANTIFICATION In order to perform label-free quantification on the NME1 dataset, target coordinates (chemical sum formulas, charge states, median retention times)
for oligonucleotides identified at 1% FDR were exported from NASE. Based on these coordinates, feature detection in the LC-MS raw data (mzML files) was carried out with the OpenMS tool
FeatureFinderMetaboIdent. The results were exported to text format using OpenMS’ TextExporter, for subsequent processing and visualization in R 3.5.150. Results from both NME1 samples were
merged and feature intensities for oligonucleotides were summed up over multiple charge and adduct states, where available. To equalize differences between replicates and ensure
comparability between conditions, the quantities for oligonucleotides that differed only in the localization of a cytidine methylation, as well as for the overlapping oligonucleotides
“UAACCCAAUGp” and “UCACAAAUCGp” (and their variants), were aggregated. REPORTING SUMMARY Further information on research design is available in the Nature Research Reporting Summary linked
to this article. DATA AVAILABILITY Mass spectrometry data files and search results (as well as label-free quantification results for the “NME1” dataset) were deposited in the PRIDE51
repository with dataset identifiers PXD012094 [https://www.ebi.ac.uk/pride/archive/projects/PXD012094] (synthetic let-7), PXD016308 [https://www.ebi.ac.uk/pride/archive/projects/PXD016308]
(NME1), PXD016323 [https://www.ebi.ac.uk/pride/archive/projects/PXD016323] (human rRNA) and PXD016328 [https://www.ebi.ac.uk/pride/archive/projects/PXD016328] (human tRNA). CODE AVAILABILITY
Source code for OpenMS, including NASE, is available on GitHub (https://github.com/OpenMS/OpenMS) under a three-clause BSD license. R scripts for post-processing of NASE results are
available by request. REFERENCES * Borek, E. & Srinivasan, P. R. The methylation of nucleic acids. _Annu. Rev. Biochem._ 35, 275–298 (1966). Article CAS Google Scholar * Davis, F. F.
& Allen, F. W. Ribonucleic acids from yeast which contain a fifth nucleotide. _J. Biol. Chem._ 227, 907–915 (1957). CAS PubMed Google Scholar * Bertero, A. et al. The SMAD2/3
interactome reveals that TGFβ controls m6A mRNA methylation in pluripotency. _Nature_ 555, 256–259 (2018). Article ADS CAS PubMed PubMed Central Google Scholar * Kirino, Y. et al.
Codon-specific translational defect caused by a wobble modification deficiency in mutant tRNA from a human mitochondrial disease. _Proc. Natl Acad. Sci. USA_ 101, 15070–15075 (2004). Article
ADS CAS PubMed PubMed Central Google Scholar * Jia, G., Fu, Y. & He, C. Reversible RNA adenosine methylation in biological regulation. _Trends Genet._ 29, 108–115 (2013). Article
CAS PubMed Google Scholar * Weng, Y.-L. et al. Epitranscriptomic m 6 a regulation of axon regeneration in the adult mammalian nervous system. _Neuron_ 97, 313–325.e6 (2018). Article
CAS PubMed PubMed Central Google Scholar * Blanco, S. et al. Aberrant methylation of tRNAs links cellular stress to neuro-developmental disorders. _EMBO J._ 33, 2020–2039 (2014). Article
CAS PubMed PubMed Central Google Scholar * Li, X., Xiong, X. & Yi, C. Epitranscriptome sequencing technologies: decoding RNA modifications. _Nat. Methods_ 14, 23–31 (2016). Article
PubMed CAS Google Scholar * Helm, M. & Motorin, Y. Detecting RNA modifications in the epitranscriptome: predict and validate. _Nat. Rev. Genet._ 18, 275–291 (2017). Article CAS
PubMed Google Scholar * Linder, B. et al. Single-nucleotide-resolution mapping of m6A and m6Am throughout the transcriptome. _Nat. Methods_ 12, 767–772 (2015). Article CAS PubMed PubMed
Central Google Scholar * Vilfan, I. D. et al. Analysis of RNA base modification and structural rearrangement by single-molecule real-time detection of reverse transcription. _J.
Nanobiotechnol._ 11, 8 (2013). Article CAS Google Scholar * Smith, A. M., Jain, M., Mulroney, L., Garalde, D. R. & Akeson, M. Reading canonical and modified nucleobases in 16S
ribosomal RNA using nanopore native RNA sequencing. _PLoS ONE_ 14, e0216709 (2019). Article CAS PubMed PubMed Central Google Scholar * Rhoads, A. & Au, K. F. PacBio sequencing and
its applications. _Genomics Proteom. Bioinforma._ 13, 278–289 (2015). Article Google Scholar * Dominissini, D. et al. The dynamic N1-methyladenosine methylome in eukaryotic messenger RNA.
_Nature_ 530, 441–446 (2016). Article ADS CAS PubMed PubMed Central Google Scholar * Safra, M. et al. The m1A landscape on cytosolic and mitochondrial mRNA at single-base resolution.
_Nature_ 551, 251–255 (2017). Article ADS CAS PubMed Google Scholar * Su, D. et al. Quantitative analysis of ribonucleoside modifications in tRNA by HPLC-coupled mass spectrometry.
_Nat. Protoc._ 9, 828–841 (2014). Article CAS PubMed PubMed Central Google Scholar * Yu, B. & Chen, X. Analysis of miRNA Modifications. _Methods Mol. Biol._ 592, 137–148 (2010).
Article CAS PubMed PubMed Central Google Scholar * Kullolli, M., Knouf, E., Arampatzidou, M., Tewari, M. & Pitteri, S. J. Intact MicroRNA ametry. _J. Am. Soc. Mass. Spectrom._ 25,
80–87 (2014). Article ADS CAS PubMed Google Scholar * Huber, C. G. & Oberacher, H. Analysis of nucleic acids by on-line liquid chromatography–mass spectrometry. _Mass. Spectrom.
Rev._ 20, 310–343 (2001). Article ADS CAS PubMed Google Scholar * Rozenski, J. & McCloskey, J. A. SOS: a simple interactive program for ab initio oligonucleotide sequencing by mass
spectrometry. _J. Am. Soc. Mass. Spectrom._ 13, 200–203 (2002). Article CAS PubMed Google Scholar * Nakayama, H. et al. Ariadne: a database search engine for identification and chemical
analysis of RNA using tandem mass spectrometry data. _Nucleic Acids Res._ 37, e47–e47 (2009). Article MathSciNet PubMed PubMed Central CAS Google Scholar * Nyakas, A., Blum, L. C.,
Stucki, S. R., Reymond, J.-L. & Schürch, S. OMA and OPA—software-supported mass spectra analysis of native and modified nucleic acids. _J. Am. Soc. Mass. Spectrom._ 24, 249–256 (2012).
Article PubMed CAS Google Scholar * Yu, N., Lobue, P. A., Cao, X. & Limbach, P. A. RNAModMapper: RNA modification mapping software for analysis of liquid chromatography tandem mass
spectrometry data. _Anal. Chem._ 89, 10744–10752 (2017). Article CAS PubMed Google Scholar * Gillet, L. C., Leitner, A. & Aebersold, R. Mass spectrometry applied to bottom-up
proteomics: entering the high-throughput era for hypothesis testing. _Annu. Rev. Anal. Chem._ 9, 449–472 (2016). Article Google Scholar * Röst, H. L. et al. OpenMS: a flexible open-source
software platform for mass spectrometry data analysis. _Nat. Methods_ 13, 741–748 (2016). Article PubMed CAS Google Scholar * Kohlbacher, O. et al. TOPP—the OpenMS proteomics pipeline.
_Bioinforma_ 23, e191–e197 (2007). Article CAS Google Scholar * Sturm, M. & Kohlbacher, O. TOPPView: an open-source viewer for mass spectrometry data. _J. Proteome Res._ 8, 3760–3763
(2009). Article CAS PubMed Google Scholar * Junker, J. et al. TOPPAS: a graphical workflow editor for the analysis of high-throughput proteomics data. _J. Proteome Res._ 11, 3914–3920
(2012). Article CAS PubMed Google Scholar * Mcluckey, S. A., Berkel, G. J. & Glish, G. L. Tandem mass spectrometry of small, multiply charged oligonucleotides. _J. Am. Soc. Mass.
Spectrom._ 3, 60–70 (1992). Article CAS PubMed Google Scholar * Lobue, P. A., Yu, N., Jora, M., Abernathy, S. & Limbach, P. A. Improved application of RNAModMapper – an RNA
modification mapping software tool – for analysis of liquid chromatography tandem mass spectrometry (LC-MS/MS) data. _Methods_ 156, 128–138 (2019). Article CAS PubMed Google Scholar *
Cox, J. & Mann, M. MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. _Nat. Biotechnol._ 26,
1367–1372 (2008). Article CAS PubMed Google Scholar * Taoka, M. et al. Landscape of the complete RNA chemical modifications in the human 80S ribosome. _Nucleic Acids Res._ 46, 9289–9298
(2018). Article CAS PubMed PubMed Central Google Scholar * Pan, T. Modifications and functional genomics of human transfer RNA. _Cell Res._ 28, 395–404 (2018). Article CAS PubMed
PubMed Central Google Scholar * Machnicka, M. A., Olchowik, A., Grosjean, H. & Bujnicki, J. M. Distribution and frequencies of post-transcriptional modifications in tRNAs. _RNA Biol._
11, 1619–1629 (2015). Article PubMed Central Google Scholar * Jühling, F. et al. tRNAdb 2009: compilation of tRNA sequences and tRNA genes. _Nucleic Acids Res._ 37, D159–D162 (2009).
Article PubMed CAS Google Scholar * de Crécy-Lagard, V. et al. Matching tRNA modifications in humans to their known and predicted enzymes. _Nucleic Acids Res._ 47, 2143–2159 (2019).
Article PubMed PubMed Central CAS Google Scholar * Rapino, F. et al. Codon-specific translation reprogramming promotes resistance to targeted therapy. _Nature_ 558, 605–609 (2018).
Article ADS CAS PubMed Google Scholar * Roundtree, I. A., Evans, M. E., Pan, T. & He, C. Dynamic RNA modifications in gene expression regulation. _Cell_ 169, 1187–1200 (2017).
Article CAS PubMed PubMed Central Google Scholar * Reinhart, B. J. et al. The 21-nucleotide let-7 RNA regulates developmental timing in Caenorhabditis elegans. _Nature_ 403, 901–906
(2000). Article ADS CAS PubMed Google Scholar * Motorin, Y. & Grosjean, H. Multisite-specific tRNA:m5C-methyltransferase (Trm4) in yeast Saccharomyces cerevisiae: identification of
the gene and substrate specificity of the enzyme. _RNA_ 5, 1105–1118 (1999). Article CAS PubMed PubMed Central Google Scholar * Cai, W. M. et al. A platform for discovery and
quantification of modified ribonucleosides in RNA: application to stress-induced reprogramming of trna modifications. _Methods Enzymol._ 560, 29–71 (2015). Article CAS PubMed PubMed
Central Google Scholar * Boccaletto, P. et al. MODOMICS: a database of RNA modification pathways. 2017 update. _Nucleic Acids Res._ 46, D303–D307 (2018). Article CAS PubMed Google
Scholar * Fenyö, D. & Beavis, R. C. A method for assessing the statistical significance of mass spectrometry-based protein identifications using general scoring schemes. _Anal. Chem._
75, 768–774 (2003). Article PubMed CAS Google Scholar * Käll, L., Storey, J. D., MacCoss, M. J. & Noble, W. S. Assigning significance to peptides identified by tandem mass
spectrometry using decoy databases. _J. Proteome Res._ 7, 29–34 (2008). Article PubMed CAS Google Scholar * Griss, J. et al. The mzTab data exchange format: communicating
mass-spectrometry-based proteomics and metabolomics experimental results to a wider audience. _Mol. Cell. Proteom._ 13, 2765–2775 (2014). Article CAS Google Scholar * Weisser, H. &
Choudhary, J. S. Targeted feature detection for data-dependent shotgun proteomics. _J. Proteome Res._ 16, 2964–2974 (2017). Article CAS PubMed PubMed Central Google Scholar * Martens,
L. et al. mzML—a community standard for mass spectrometry data. _Mol. Cell. Proteom._ 10, R110.000133 (2011). Article Google Scholar * Chambers, M. C. et al. A cross-platform toolkit for
mass spectrometry and proteomics. _Nat. Biotechnol._ 30, 918–920 (2012). Article CAS PubMed PubMed Central Google Scholar * Taoka, M. et al. The complete chemical structure of
_Saccharomyces cerevisiae_ rRNA: partial pseudouridylation of U2345 in 25S rRNA by snoRNA snR9. _Nucleic Acids Res._ 44, 8951–8961 (2016). Article CAS PubMed PubMed Central Google
Scholar * R Core Team. _R: A language and environment for statistical computing_. (R Foundation for Statistical Computing, 2018). * Vizcaíno, J. A. et al. 2016 update of the PRIDE database
and its related tools. _Nucleic Acids Res._ 44, 11033 (2016). Article PubMed PubMed Central CAS Google Scholar Download references ACKNOWLEDGEMENTS We would like to thank the following
people: Lina Vasiliauskaitė and Alan Hendrick for preparing the HAP1 tRNA and rRNA samples, respectively. Jack Rogan for performing the RNA fractionation during rRNA sample generation.
Ningxi Yu for suggesting optimal parameters for running his RNAModMapper tool. Hiroshi Nakayama for sharing the TK6 rRNA sequences used by Taoka et al.32. All contributors to OpenMS;
especially Hannes Röst for his efforts and useful feedback during the code review for this project. B.A.G. acknowledges funding from NIH grant GM110174 and a UPenn Epigenetics Institute
Pilot grant. AUTHOR INFORMATION Author notes * These authors contributed equally: Samuel Wein, Byron Andrews. AUTHORS AND AFFILIATIONS * Epigenetics Program, Perelman School of Medicine,
University of Pennsylvania, Philadelphia, PA, USA Samuel Wein & Benjamin A. Garcia * Center for Bioinformatics Tübingen, University of Tübingen, Tübingen, Germany Samuel Wein &
Oliver Kohlbacher * STORM Therapeutics Limited, Moneta Building, Babraham Research Campus, Cambridge, UK Byron Andrews & Hendrik Weisser * Applied Bioinformatics, Department for Computer
Science, University of Tübingen, Tübingen, Germany Timo Sachsenberg & Oliver Kohlbacher * Gurdon Institute, University of Cambridge, Cambridge, UK Helena Santos-Rosa & Tony
Kouzarides * Quantitative Biology Center, University of Tübingen, Tübingen, Germany Oliver Kohlbacher * Biomolecular Interactions, Max Planck Institute for Developmental Biology, Tübingen,
Germany Oliver Kohlbacher * Translational Bioinformatics, University Hospital Tübingen, Tübingen, Germany Oliver Kohlbacher Authors * Samuel Wein View author publications You can also search
for this author inPubMed Google Scholar * Byron Andrews View author publications You can also search for this author inPubMed Google Scholar * Timo Sachsenberg View author publications You
can also search for this author inPubMed Google Scholar * Helena Santos-Rosa View author publications You can also search for this author inPubMed Google Scholar * Oliver Kohlbacher View
author publications You can also search for this author inPubMed Google Scholar * Tony Kouzarides View author publications You can also search for this author inPubMed Google Scholar *
Benjamin A. Garcia View author publications You can also search for this author inPubMed Google Scholar * Hendrik Weisser View author publications You can also search for this author
inPubMed Google Scholar CONTRIBUTIONS S.W., T.S., and H.W. developed the software. S.W., B.A., and H.W. analyzed the data and wrote the paper. B.A. performed the LC-MS/MS experiments. H.S-R.
prepared the NME1 samples. O.K., T.K., and B.A.G. provided resources and high-level supervision. All authors read and approved the manuscript. CORRESPONDING AUTHORS Correspondence to
Benjamin A. Garcia or Hendrik Weisser. ETHICS DECLARATIONS COMPETING INTERESTS T.K. is a founder and director of STORM Therapeutics Limited, Cambridge, UK. B.A. and H.W. are full-time
employees of STORM Therapeutics Limited, Cambridge, UK. S.W., T.S., H.S-R., O.K. and B.A.G. declare no competing interests. ADDITIONAL INFORMATION PEER REVIEW INFORMATION _Nature
Communications_ thanks the anonymous reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available. PUBLISHER’S NOTE Springer Nature remains neutral
with regard to jurisdictional claims in published maps and institutional affiliations. SUPPLEMENTARY INFORMATION SUPPLEMENTARY INFORMATION PEER REVIEW FILE DESCRIPTION OF ADDITIONAL
SUPPLEMENTARY FILES SUPPLEMENTARY DATA 1 REPORTING SUMMARY RIGHTS AND PERMISSIONS OPEN ACCESS This article is licensed under a Creative Commons Attribution 4.0 International License, which
permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to
the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless
indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or
exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/. Reprints
and permissions ABOUT THIS ARTICLE CITE THIS ARTICLE Wein, S., Andrews, B., Sachsenberg, T. _et al._ A computational platform for high-throughput analysis of RNA sequences and modifications
by mass spectrometry. _Nat Commun_ 11, 926 (2020). https://doi.org/10.1038/s41467-020-14665-7 Download citation * Received: 11 February 2019 * Accepted: 22 January 2020 * Published: 17
February 2020 * DOI: https://doi.org/10.1038/s41467-020-14665-7 SHARE THIS ARTICLE Anyone you share the following link with will be able to read this content: Get shareable link Sorry, a
shareable link is not currently available for this article. Copy to clipboard Provided by the Springer Nature SharedIt content-sharing initiative
